Educating a New Generation of Human-Centered Designers for the Medical Device Industry
Futures, Bold & Curious at NCAD
Email: postgraduate@ncad.ie
The Development of MSc Medical Device Design at NCAD
The roots of the program date to the 1980s when U.S. multinationals like Baxter and CR Bard established European manufacturing bases in Ireland to take advantage of an educated workforce, cheap labor and incentives from the Irish Industrial Development Authority. Since then, Ireland’s economy has advanced. High levels of education have enabled a strong research sector and development in both industry and academia. Among the more than 300 companies with Irish facilities today are Boston Scientific, Abbott, Vistakon, Medtronic, Teleflex, Stryker, Cook Medical, Zimmer Biomet, DePuy Synthes, Hollister, and BD.
In 2009, NCAD’s head of industrial design realized that graduates were being hired in the medical device industry. He saw a need for designers in the medical device industry who married design’s human-focused approach with the rigor of science. Working with University College Dublin and Trinity College Dublin, the MSc in Medical Device Design was created with classes that include medical science and bioengineering, alongside studio projects undertaken in collaboration with industry.

Bridging Design and Technology Students come to the program with either a technical background or a design background, and on occasion both. While designers can be too conceptual and emphasize aesthetics, engineers can be technology driven and fail to adequately consider users. The MSc program seeks to remedy these tendencies by teaching an iterative process of analysis and synthesis in an open studio environment. Students are encouraged to share ideas but also must confront the technically rigorous aspects of medical design.
NCAD's Human-Centred Approach
The aim of the program is to produce medical designers who apply human-factor methods as an integral part of the design process. Designers need to be a voice of empathy for the user, clinician, patient and caregiver. Until recently, many engineers and designers were discouraged from visiting the clinical environment, which was thought to be the domain of market research or product managers. Conducting contextual inquiry and mapping tasks and user journeys are becoming vital skills for both designers and engineers. Many bigger design teams may have a human factors specialist who can diagnose errors and pain points in a device but may not be skilled in elaborating solutions. A designer with the right training can use the insights gained through contextual inquiry to inspire novel designs and improvements. The designer can use sketches and sketch models to facilitate conversation and collaboration among engineers, designers, clinicians and patient advocates in a multidisciplinary team.
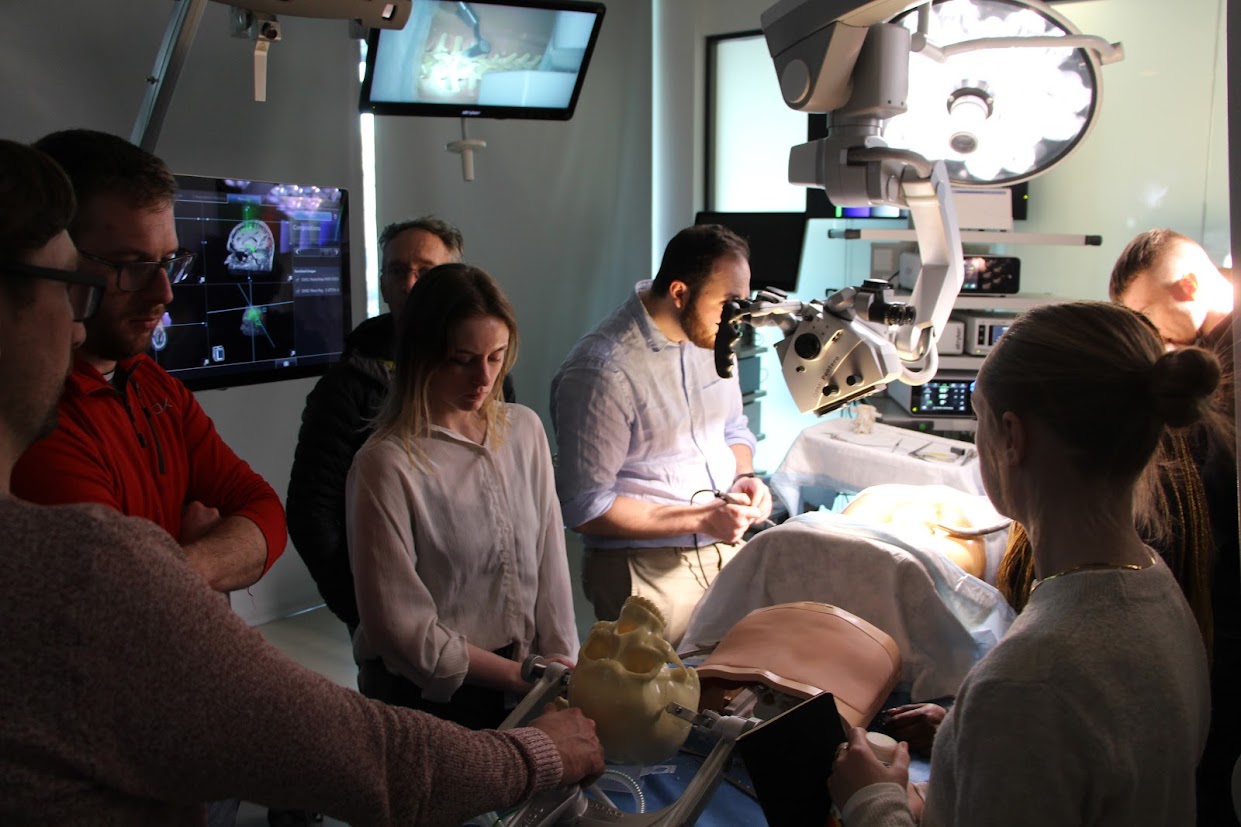
All project work in the studio is conducted with realworld collaborators, be they medical device companies, local hospitals or medical schools. Each year, the students’ first project, in collaboration with the Royal College of Surgeons, involves building a task trainer on which trainee surgeons can practice a procedure. The students begin with conducting primary and secondary research and mapping out the task in great detail. Capturing the perceptions, cognitions and actions of a surgeon as they execute a procedure is no simple exercise, and building a device to simulate the same is harder still. Concurrent with the project work, the students have classes in human factors, materials, and failure modes and effects analysis, as well as basic medical science at Trinity College Dublin. Students also work on industry projects that call for creative problemsolving rooted contextual inquiry. The students work with medical device multinationals and indigenous startups to produce design concepts based on research conducted with local hospitals. These industry collaborators value fresh thinking, and many of the ideas developed in the program have been patented over the years.
Working in Medical Device Industry
Design in medical devices tends to evolve more slowly than in other areas. What is considered a major innovation in this area would be seen as an incremental improvement in another specialization. This is mainly due to regulatory constraints and the unique economic situation of the medical sector. With medical devices you must consider who will choose, use and pay the dues for a device. Usually these are three distinct entities. Normal laws of supply and demand are distorted by factors such as reimbursement codes and health insurance.
Students get a sense of these factors in two week-long projects with local hospitals. In each, an open call is put out to hospital staff to work with the students for the week to solve a problem in the hospital. In the Mater Misericordiae Hospital, the MSc students work alongside students from the MA Interaction Design and MA Service Design programs to improve anything from wayfinding to information sharing. In St. James’s Hospital, they collaborate with bioengineering students from Trinity College Dublin. Experience in a hospital is a valuable chance to build empathy with the staff, patients and caregivers who will use your designs. It also teaches the complexity of medical care. For patients, caregivers and clinicians, it is often simple things that matter. Recognizing these details is a vital part of the designer’s job.
In the second semester, students have classes and laboratories in bioinstrumentation with the medical physics team at St. James’s Hospital. This is an opportunity to learn about the complex science of human measurement that interacts with so many medical devices. It is an opportunity to look for inspiration for new devices. How might we incorporate sensors or measurements into a device? It is also a chance to look toward a utopian future beyond the device. How might we measure these vital signs before the condition develops and before the user becomes a patient?
On the Horizon Recognizing the importance of new technology and all the interaction points along a patient journey means that students are not afraid of the messier spaces that many of the latest innovations in healthcare end up in. Medical device companies along with the engineers and scientists who work in them are experts in the physical device they currently produce but often struggle with innovation beyond this space.

The medical industry is at varying levels of advancement. While practitioners are always interested in the newest technologies, some institutions worldwide still use faxes to communicate. Efforts to create an accessible patient record system have fallen foul of bureaucracy and the market, where those who are developing the technology are unwilling or unable to allow the different record databases to talk to one another. A world where patients have access to their own data is still in the distance. Designing patient records in a manner that is both accessible to the patient and not a frightening experience for them will be a job for designers of the future.
The medical device industry has come late to sustainability, and for many companies, “single-use” is a badge of honor for preventing the spread of infection. In the last few years, device companies have started talking about sustainability as a market advantage. End users have begun questioning the costs involved in the disposal of biomedical waste. For example, the rise of cheap consumer electronics has led to the development of singleuse chip-on-tip endoscopes; however, not all clinicians are comfortable throwing away a device that contains nonrenewable materials, many of which are on the critical list. As a result, a recent graduate of the MSc program has founded a startup to design an innovative endoscope for the circular economy. This company has received funding for a collaboration with the college to develop the device. This will require that the device be returned to the producer, disassembled, sterilized and still have the same safety and efficacy it had the first time around. This is a complex threeyear project bringing together optoelectronics, medical device design and sustainability.
The increasing importance of human factors in the medical device industry is often seen as another regulatory hurdle, but progressive companies are starting to see design and human factors research as a rich space for innovation, intellectual property and product differentiation. A future where data and automation enhance medical care will require well-trained human-focused designers who are not afraid of the spaces in-between. The designer’s job in this complex ecosystem is to ensure that the development team never forgets the user and to design the device’s physical and digital touchpoints in accordance with the user’s needs. Exactly the type of designer NCAD’s MSc program Medical Device Design is empowering.
Enda O’Dowd, I/IDSA, and Derek Vallence, I/IDSA odowde@staff.ncad.ie; vallenced@staff.ncad.ie
Enda O’Dowd
Enda O'Dowd is a lecturer in the Product Design Department and joint course coordinator of the MSc Medical Device Design. Enda’s background is in materials and polymer technology and he has pos
Derek Vallence BDes., MSc.
Derek i s a lecturer in the Product Design Department and joint course coordinator of the MSc Medical Device Design. Derek holds a BDes in Industrial Design and an MSc in Computer Aided Product Design. His background is in 3D modelling, simulation, prototyping and design for manufacture. On the MSc course, along with supervising projects with industry, he also teaches CAD along with a series of lectures on human factors tools for usability testing. His area of interest and research is in Design for Health and the Democratisation of Manufacturing processes.
s a lecturer in the Product Design Department and joint course coordinator of the MSc Medical Device Design. Derek holds a BDes in Industrial Design and an MSc in Computer Aided Product Design. His background is in 3D modelling, simulation, prototyping and design for manufacture. On the MSc course, along with supervising projects with industry, he also teaches CAD along with a series of lectures on human factors tools for usability testing. His area of interest and research is in Design for Health and the Democratisation of Manufacturing processes.
For further information on how to apply for MSc Medical Device Design join our webinar on 28 Feb 4pm.